

The integration of internet-connected medical devices into patient care environments has reached a point where a single compromised thermostat can jeopardize the safety of an entire surgical department. While the protection of electronic health records was once the primary focus for cybersecurity
For individuals residing in the more isolated stretches of the regional landscape, the traditional model of specialized disability support frequently involved insurmountable travel times and long waitlists for visiting practitioners. By 2026, the National Disability Insurance Scheme has undergone a

The traditional bathroom scale has long functioned as a source of morning anxiety rather than a tool for clinical insight, yet the recent launch of the Withings BodyFit represents a fundamental shift toward comprehensive physiological monitoring. By moving beyond the singular metric of total body
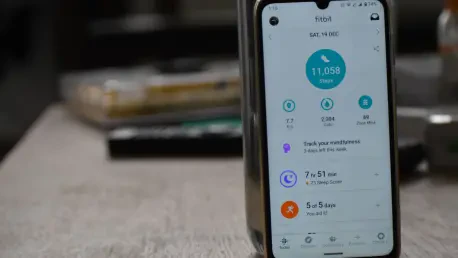
The rapid evolution of generative artificial intelligence has fundamentally altered the landscape of consumer electronics, moving wearables from simple pedometers to sophisticated diagnostic tools. Samsung has positioned its latest software suite at the forefront of this shift, integrating complex

The healthcare technology sector is currently facing an unprecedented wave of regulatory hurdles as oversight bodies significantly increase the frequency of rejections for medical device submissions that do not meet rigorous cybersecurity criteria. This trend reflects a broader shift in the
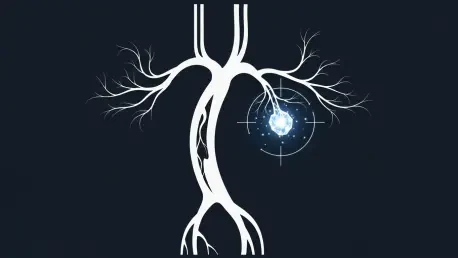
The landscape of vascular medicine underwent a radical shift when clinical researchers introduced a molecular imaging technique capable of mapping every thrombus in the human body simultaneously. For decades, medical professionals relied on fragmented snapshots provided by disparate imaging

The healthcare landscape recently encountered a significant bottleneck as the Federal Independent Dispute Resolution process struggled to keep pace with the massive volume of payment disagreements triggered by the No Surprises Act. This landmark legislation, while highly successful in shielding

Modern healthcare facilities are grappling with an unprecedented complexity in medication management that demands both mechanical precision and specialized human expertise to ensure patient safety. As pharmacists and technicians navigate high-pressure environments, the margin for error remains
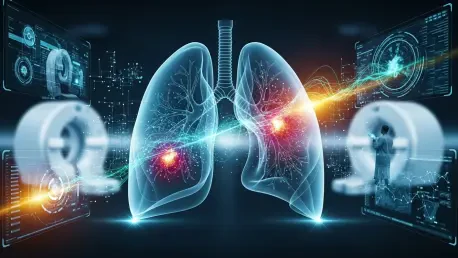
The diagnostic landscape for pulmonary disorders has reached a critical juncture where traditional imaging modalities often fail to capture the physiological nuances of gas exchange and regional blood flow required for precision medicine. While computed tomography remains the gold standard for
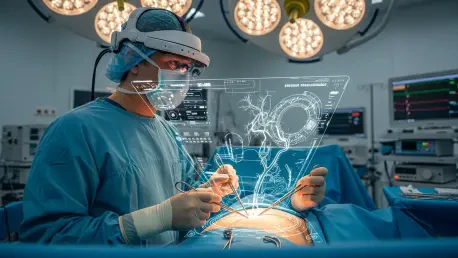
Medical professionals operating in complex anatomical environments have traditionally relied on their internal mental mapping to translate two-dimensional preoperative scans into the three-dimensional reality of a living patient. This cognitive load often introduces a margin for error, particularly
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59