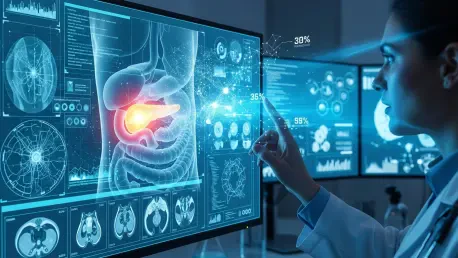
The medical community has long struggled with the silent progression of pancreatic ductal adenocarcinoma, a disease that frequently escapes detection until it reaches its most lethal and untreatable stages. Current diagnostic protocols typically rely on manual review of computed tomography scans, yet even the most experienced radiologists often fail to identify subtle structural changes in the early phases of malignancy. Because over 85 percent of patients present with advanced-stage tumors that are ineligible for curative surgery, the five-year survival rate remains one of the lowest in oncology. Recent breakthroughs in artificial intelligence offer a promising shift in this paradigm, specifically through a newly developed framework known as the Radiomics-based Early Detection Model, or REDMOD. This technology leverages deep learning to identify what experts call visually occult disease, pinpointing microscopic tissue alterations that precede a clinical diagnosis by months or even years, thereby creating a vital window for life-saving medical intervention.
Technological Foundations: The REDMOD Framework
The underlying architecture of this system utilizes a fully automated pipeline designed to remove the subjectivity inherent in human-led radiological assessments. Developed by researchers at the Mayo Clinic, the REDMOD framework integrates sophisticated pancreas segmentation with an advanced radiomic feature extraction process. The training phase involved a massive, multi-institutional dataset consisting of 1,462 scans, which included both prediagnostic images from future cancer patients and control images from healthy individuals. A critical discovery during this development phase was that 90 percent of the diagnostic features used by the AI were derived from wavelet-filtered images. These filters allow the software to analyze complex tissue textures that are invisible to the naked eye under standard lighting and contrast settings. By focusing on these quantitative markers, the model identifies specific patterns of irregularity that indicate the presence of a tumor long before a solid mass begins to distort the visible boundaries.
Building on this computational foundation, the AI specifically targets 40 unique quantitative markers that act as biological signatures of early-stage malignancy. These markers are not just random data points; they represent mechanistically grounded changes in the density and heterogeneity of pancreatic tissue. In standard clinical practice, a radiologist might look for a localized mass or a dilated duct, but the REDMOD system observes the subtle shift in the spatial arrangement of pixels that suggests a change in the cellular environment. This approach is particularly effective because it compensates for the common variations found in routine CT scans across different medical facilities. Whether the imaging is performed on older equipment or the latest hardware, the model maintains a high degree of sensitivity by filtering out visual noise and focusing on the core textures that signify disease. This level of granularity ensures that the diagnostic process is both robust and consistent, providing a standardized metric for evaluating patients.
Performance Metrics: Clinical Integration and Future Steps
The performance data gathered during independent testing reveals a transformative shift in the speed and accuracy of detection compared to traditional methods. While human experts achieved a pooled sensitivity of only 38.9 percent when reviewing prediagnostic scans, the REDMOD model reached an Area Under the Curve of 0.82 with a sensitivity of 73 percent. This disparity highlights the fundamental limitation of human vision in detecting the earliest stages of pancreatic ductal adenocarcinoma. Perhaps the most striking finding is the model’s ability to maintain high performance even when the imaging was conducted a year and a half before the clinical symptoms appeared. In several instances, the AI identified signs of malignancy at a median lead time of 475 days prior to a standard diagnosis. Even at the 24-month mark, the model maintained a sensitivity of 68 percent, nearly triple the effectiveness of human radiologists who reviewed the same scans during that period.
The development and successful validation of the REDMOD model marked a pivotal moment in the fight against pancreatic ductal adenocarcinoma. By shifting the diagnostic focus from visible anatomical distortions to subtle texture-based markers, the research team offered a tangible solution to the problem of late-stage diagnosis. The study confirmed that artificial intelligence could provide a lead time of over a year, granting physicians and patients the most valuable resource in oncology: time. Moving forward, the integration of these tools into standard practice provided a blueprint for how computational analysis could complement human expertise. Rather than replacing the radiologist, the technology acted as a high-precision filter that caught early signs of disease that the human eye was simply not equipped to see. This shift in methodology represented a fundamental change in how the medical community approached cancer detection, moving away from a reliance on reactive measures and toward a more data-driven strategy.