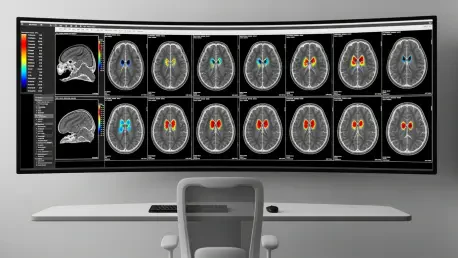
The landscape of modern neuroimaging is currently witnessing a monumental shift as a pioneering multiplexed magnetic resonance imaging technology known as MRx begins to redefine clinical diagnostics. Developed by a multidisciplinary research team at the University of Illinois Urbana-Champaign, this innovation represents a significant departure from the historical limitations of traditional medical imaging, which has long relied on tracking the distribution of water molecules to visualize internal anatomy. While standard scans excel at identifying structural abnormalities like major trauma or late-stage tumors, they often overlook the nuanced biochemical shifts that occur at the earliest stages of disease. MRx addresses this critical gap by providing a high-resolution, multidimensional view of the brain’s biochemical and physiological environment, successfully capturing more than 20 distinct biomarkers within a single, efficient session. This breakthrough allows for a comprehensive assessment of the human brain that was previously considered unattainable within a standard clinical timeframe.
The Synergy: Integrating Physics and Artificial Intelligence
The technical foundation of MRx rests upon a sophisticated fusion of hardware optimization and computational intelligence that fundamentally changes how magnetic resonance data is gathered. Historically, capturing a diverse range of biomarkers would have required multiple, separate scans, each specifically tailored to a specific molecule or metabolic process, leading to hours of equipment time that is simply not feasible in a hospital setting. To bypass this bottleneck, the research team, led by Professor Zhi-Pei Liang, developed specialized pulse sequences designed to sample a wide array of molecular signals simultaneously rather than sequentially. This method taps into the resonant frequencies of various biological molecules, including vital metabolites and neurotransmitters, effectively creating a biochemical fingerprint of the brain. By maximizing the data density of every second spent in the scanner, the technology provides a holistic view of tissue metabolism and cellular health in real-time.
To manage the inherent complexity of these overlapping signals, the system utilizes physics-based machine learning algorithms that are deeply grounded in the fundamental laws of magnetic resonance. Unlike standard “black-box” artificial intelligence models that can produce unpredictable results, these algorithms are specifically designed to untangle the interleaved signals produced by different molecular species with extreme precision. They effectively filter out background noise while separating the data into high-fidelity maps, solving a decades-old trade-off between imaging speed and spatial resolution. This computational prowess ensures that the final output remains remarkably clear and accurate, even as the acquisition speed increases to levels that match modern clinical demands. This synergy between advanced physical modeling and rapid data processing marks a turning point in the ability of clinicians to visualize the invisible chemical processes that govern neurological function.
Clinical Efficiency: Streamlining Diagnostics and Patient Safety
One of the most significant practical advantages of MRx is its remarkable speed, which allows for a comprehensive whole-brain scan mapping 21 different biomarkers to be completed in roughly 14 minutes. This duration is almost identical to standard clinical protocols for routine structural imaging, ensuring that the technology can be seamlessly integrated into busy hospital schedules without disrupting existing workflows. For patients, shorter scan times are not merely a matter of convenience; they significantly reduce the physical discomfort associated with remaining perfectly still in a confined space. This reduction in time also drastically lowers the likelihood of motion-related artifacts in the data, which often plague longer scans and lead to the need for costly and time-consuming re-imaging. By making high-end molecular diagnostics as fast as a standard anatomical check-up, the technology bridges the gap between research-grade insights and routine clinical care.
Furthermore, MRx achieves these high-resolution results without the necessity of injectable contrast agents, which has been a staple of diagnostic imaging for several decades. Many traditional procedures rely on gadolinium-based contrast media to highlight specific tissues or vascular issues, but these agents carry inherent risks, including potential toxicity for patients with compromised kidney function and the rare possibility of severe allergic reactions. By eliminating the requirement for external chemical enhancements, the MRx procedure simplifies the preparatory process for both medical staff and patients while simultaneously reducing procedural risks. This advancement makes sophisticated diagnostic imaging accessible to a far broader demographic, including those who were previously excluded from advanced scans due to underlying health conditions. The transition toward non-invasive, contrast-free molecular phenotyping represents a safer and more inclusive path forward for the entire medical imaging industry.
Precision Neurology: Transforming Oncology and Chronic Disease Management
The transformative potential of MRx is perhaps most evident in its immediate application to complex neurological disorders, particularly in the delicate field of oncology. In the study of brain tumors, this technology has demonstrated a unique ability to delineate the microenvironment of a lesion with a level of detail that traditional imaging simply cannot match. While a conventional MRI might only show a single, undifferentiated mass, MRx can distinguish between areas of active metabolic disruption, swelling, axonal injury, and demyelination within that same area. This nuance allows oncologists to differentiate between various tumor grades and states that might appear identical on a standard scan, providing critical information for surgical planning. By identifying which parts of a lesion are most aggressive or metabolically active, surgeons and radiologists can tailor their interventions with a level of precision that significantly improves patient outcomes.
Beyond the realm of cancer, this technology offers a multifaceted analysis of brain lesions in patients suffering from multiple sclerosis, which has historically been difficult to monitor in real-time. MRx can identify the specific stage of a lesion—whether it is characterized by active inflammation, scarring, or permanent nerve damage—by analyzing the specific molecular signatures present in the tissue. Perhaps most importantly, the technology is sensitive enough to detect subtle biochemical alterations that occur before a visible lesion even forms on a standard anatomical scan. This predictive capability could revolutionize how chronic neurological diseases are treated, allowing for much earlier medical interventions that could significantly slow the progression of the disease. Because the technology is compatible with existing clinical MRI hardware, it is uniquely positioned for rapid global adoption, ensuring that personalized, molecular-level neurological care becomes the standard rather than the exception.
Future Outlook: Implementing Molecular Phenotyping in Modern Practice
As the medical community begins to integrate MRx into standard diagnostic pipelines, the focus must now shift toward establishing standardized molecular baselines for various patient populations across the globe. This transition from purely anatomical imaging to multiplexed molecular phenotyping requires a collaborative effort between radiologists, data scientists, and healthcare administrators to update existing diagnostic frameworks. Medical centers should prioritize the software upgrades necessary to run these physics-based machine learning algorithms on their current hardware, as this provides a cost-effective route to upgrading their diagnostic capabilities without purchasing new scanners. Training programs for clinical staff will also be essential to ensure that the wealth of biochemical data provided by MRx is interpreted correctly and translated into actionable treatment plans for patients with neurodegenerative conditions.
Looking ahead, the long-term impact of this technology will likely be measured by its ability to accelerate the development of personalized medicine for conditions like Alzheimer’s and Parkinson’s disease. By providing a panoramic view of brain metabolism and neurotransmitter activity, MRx gives researchers a powerful tool to track how specific drugs affect the brain’s chemistry in real-time. This allows for a more dynamic approach to healthcare, where treatments can be adjusted or replaced based on the individual physiological response of each patient. The shift toward a deeper understanding of the brain’s internal environment was previously limited by technical constraints, but those barriers have now been effectively dismantled. The move toward non-invasive, high-speed molecular mapping is no longer a theoretical goal but a practical reality that will refine the precision of neurological care and enhance patient safety for the foreseeable future.