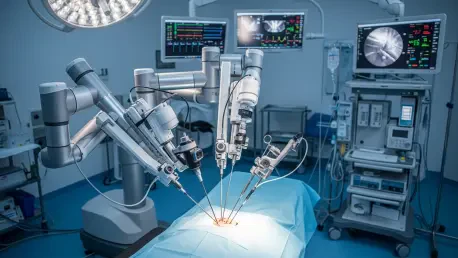
The transition from traditional factory floor automation to the ultra-sterile environment of the modern operating room represents one of the most complex engineering challenges in contemporary medical technology. Stäubli Robotics has addressed this specific demand by introducing the TX2-60L MedX Ready, a six-axis robotic arm meticulously engineered to bridge the gap between high-performance industrial motion and the stringent safety requirements of human surgery. Developed within a dedicated ISO 13485-compliant Quality Management System, this platform provides a foundational layer of reliability that allows medical equipment manufacturers to focus on patient outcomes rather than hardware limitations. By aligning with these international standards, the company ensures that the robot is optimized for hygiene and seamless integration into clinical workflows. This release signifies a departure from general-purpose machinery, offering instead a specialized solution that prioritizes clinical safety and regulatory readiness for healthcare providers worldwide.
Bridging Clinical Precision and Ergonomic Control
Central to the innovation of this robotic arm is the MedXguiding system, an intuitive manual guidance function designed to facilitate the fluid positioning of surgical instruments by clinical teams. This feature provides a natural interface where the robotic arm responds to the surgeon’s touch with high sensitivity, allowing for precise adjustments in real-time without the lag or mechanical resistance often associated with larger industrial models. The system offers various configurable modes that adapt to different surgical phases, ensuring that the workflow remains ergonomic and reduces the physical fatigue experienced by the operating staff. By streamlining the setup process and providing high rigidity during static phases, the technology ensures that instruments remain exactly where they are placed. This level of responsiveness is critical for delicate procedures in fields like neurosurgery or orthopedics, where even a fraction of a millimeter can influence the clinical success of an intervention.
The development of the TX2-60L MedX Ready is not an isolated achievement but rather the culmination of a legacy that began when the PUMA 560 robot assisted in the world’s first documented robotic neurosurgical biopsy. In the decades since, the industry has seen a massive expansion in how robotics are utilized, and Stäubli has maintained its position as a pioneer through strategic collaborations with medical technology leaders. Companies like Zimmer Biomet and Quantum Surgical have integrated these robotic foundations into advanced systems for oncology and orthopedics, demonstrating the versatility of the six-axis design. This historical expertise provides a level of confidence for hospitals adopting the technology, knowing that the platform is built on years of research and practical clinical application. The current iteration supports a broad spectrum of medical needs, from high-definition imaging support to the physical execution of complex bone cuts, proving that the hardware can meet the diverse demands of a modern surgical suite.
Streamlining Regulatory Pathways and Safety Standards
One of the most significant barriers for medical device manufacturers today is the arduous process of securing regulatory certification, a task that often extends the timeline for bringing life-saving technologies to market. Stäubli addresses this bottleneck by providing a comprehensive Validation and Documentation package with every robotic unit, encompassing everything from component traceability to detailed inspection reports. This documentation acts as a pre-verified foundation, allowing integrators to move through the auditing process with greater speed and transparency. By including production checklists and rigorous testing data, the package reduces the burden on individual manufacturers to prove the safety of the underlying motion system. This proactive approach to documentation ensures that the integration of robotics into new medical systems is less a gamble and more a streamlined transition. Such meticulous attention to administrative detail mirrors the physical precision of the arm itself, creating a holistic product that serves both engineers and regulators.
To maintain the high standards of cleanliness required for surgical environments, the robotic arm features a smooth, encapsulated design that eliminates areas where pathogens could potentially accumulate. The surfaces are resistant to aggressive cleaning agents, ensuring that the unit can be sterilized repeatedly between procedures without degrading the external materials or internal electronics. This focus on “cleanable platform” design allows medical innovators to deploy the arm in direct proximity to patients during sensitive procedures, such as robotic-assisted biopsies or targeted radiation therapy. Furthermore, the high connectivity of the TX2-60L ensures that it can communicate seamlessly with other operating room systems, creating a synchronized environment where data and motion work in harmony. By prioritizing these safety-first principles, the technology provides a stable and predictable tool for surgeons, allowing them to perform at their peak while the robotic system handles the repetitive and high-precision aspects of the surgery with absolute consistency.
Future Considerations: Strategic Implementation and Insights
The deployment of the TX2-60L MedX Ready offered a clear pathway for medical facilities to adopt robotic assistance without the traditional risks associated with non-specialized hardware. Hospital administrators and clinical engineers prioritized the selection of platforms that featured built-in regulatory compliance, as this significantly lowered the long-term costs of maintenance and recertification. The focus shifted toward modular integration, where the robotic arm served as a reliable chassis for a variety of specialized surgical tools, ranging from laser scalpels to ultrasonic probes. This adaptability ensured that the investment in robotic infrastructure remained relevant across different medical departments, maximizing the utility of the hardware. Strategic planning emphasized the importance of training clinical staff on the manual guidance features early in the adoption phase to ensure that the transition to robotic-assisted workflows remained smooth and collaborative.
Moving forward, the industry benefited from a shift toward standardized robotic documentation, which simplified the global distribution of advanced surgical systems. Manufacturers who utilized the pre-validated documentation packages saw a reduction in time-to-market, allowing them to respond more quickly to emerging clinical needs in areas like oncology and minimally invasive surgery. The integration of high-rigidity robotics also paved the way for more complex outpatient procedures, as the precision of the TX2-60L MedX Ready allowed for smaller incisions and faster recovery times for patients. Future considerations for developers included the expansion of haptic feedback systems to further enhance the tactile connection between the surgeon and the robot. By providing a clean, compliant, and highly communicative foundation, the platform established a new benchmark for how industrial-grade engineering could be successfully repurposed for the most demanding applications in human healthcare.