
Modern healthcare facilities are grappling with an unprecedented complexity in medication management that demands both mechanical precision and specialized human expertise to ensure patient safety. As pharmacists and technicians navigate high-pressure environments, the margin for error remains
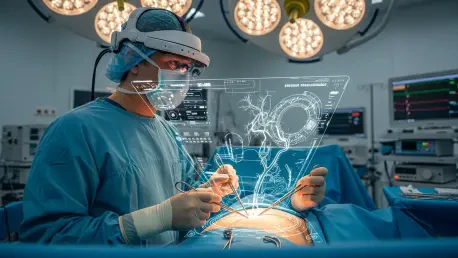
Medical professionals operating in complex anatomical environments have traditionally relied on their internal mental mapping to translate two-dimensional preoperative scans into the three-dimensional reality of a living patient. This cognitive load often introduces a margin for error, particularly
The recent surge in complex cardiac interventions has placed a massive spotlight on the mechanical assist devices that sustain life when the human heart falters during surgery. The acquisition of Abiomed for $16.6 billion signaled Johnson & Johnson’s aggressive expansion into high-growth medtech

The successful completion of a major fundraising campaign often marks a transformative moment for regional healthcare facilities as they strive to integrate sophisticated surgical technologies into their oncology departments. Victoria Hospital recently announced that it surpassed its ambitious

The escalating prevalence of sophisticated exploitation within the durable medical equipment sector has prompted a significant legislative response aimed at preserving the financial integrity of the United States healthcare infrastructure. On May 21, 2026, the House Ways and Means Committee
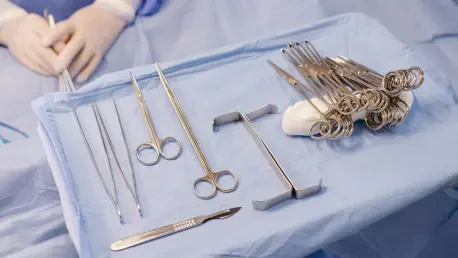
The sudden realization that surgical instruments have not undergone the rigorous sterilization protocols required for invasive procedures represents a catastrophic breakdown in hospital safety management. For sixty-eight-year-old Sally-Ann Pyrah-Barnes, this nightmare became a reality at the St.