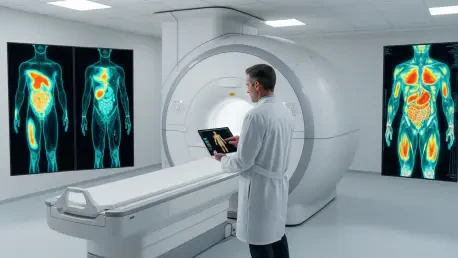
The long-standing reliance on the Body Mass Index as a definitive measure of human health is rapidly unraveling as modern medical technology reveals the hidden complexities of internal physiology. While clinicians have used the ratio of weight to height for decades to categorize patients, this metric remains fundamentally flawed because it fails to distinguish between heavy muscle and dangerous internal fat. A groundbreaking shift is now occurring through the application of artificial intelligence to whole-body magnetic resonance imaging, allowing for a level of anatomical scrutiny previously deemed impossible at scale. This technological leap, recently highlighted in a major study, demonstrates how deep-learning frameworks can map a person’s internal landscape to predict long-term risks for chronic conditions like diabetes and heart disease. By looking past the surface level of the scale, this new approach provides a quantitative assessment of health that prioritizes where tissue is located rather than simply how much a person weighs in total.
Analyzing Population Scales through Automated Pipelines
The implementation of this advanced diagnostic approach required the analysis of a massive dataset consisting of over 66,000 participants from major longitudinal studies across Europe. Researchers utilized a fully automated deep-learning segmentation pipeline to process an immense volume of imaging data, which previously would have required thousands of hours of manual labor by expert radiologists. This AI-driven framework was specifically designed to identify and measure five distinct body composition metrics, including visceral adipose tissue, which surrounds internal organs, and skeletal muscle fat fraction. By tracking these participants over a period that has extended into the current year, the study established a robust baseline for how internal tissue changes over time. This level of technical rigor allowed the researchers to move beyond the “overweight” or “obese” labels provided by traditional metrics, identifying deeper physiological markers that often remain invisible during standard checkups.
Beyond the sheer volume of data, the precision of the segmentation process represents a major technical achievement in the field of medical imaging. The AI models were trained to recognize the subtle differences between subcutaneous fat, which sits just under the skin, and intramuscular adipose tissue, which infiltrates the muscle fibers like marbling in meat. This distinction is critical because different types of fat carry vastly different metabolic consequences for the individual. The automated nature of this pipeline ensures that such detailed analysis can be performed consistently across tens of thousands of scans, removing the subjectivity that often accompanies manual image interpretation. As these tools become more refined, the ability to generate a complete map of a person’s metabolic health in seconds will likely transform how radiologists and general practitioners collaborate on preventative care strategies, shifting the focus from treating existing illnesses to identifying risks before symptoms even emerge.
Establishing Global Standards with Normative Reference Maps
To translate these complex imaging findings into something a primary care physician can use, the researchers developed a series of normative reference maps. These maps act as a standardized yardstick, accounting for the natural variations in body composition that occur due to a person’s age, biological sex, and height. By converting raw tissue volumes into standardized “z-scores,” the system can categorize individuals into low, middle, or high-risk groups relative to their demographic peers. This transition from absolute volume to a relative risk score is a vital step in making imaging biomarkers practical for everyday medicine. It ensures that a patient’s results are interpreted within a meaningful context, preventing a naturally muscular individual from being misclassified as high-risk. This methodology creates a personalized health profile that reflects the unique physiological reality of each patient, providing a much clearer picture of their metabolic state.
The utility of these standardized maps extends into the realm of long-term epidemiological modeling and clinical workflow integration. When a doctor views a patient’s z-score, they are not just looking at a number; they are seeing a representation of that patient’s position within a vast population of similar individuals. This allows for a more nuanced conversation about health, where the focus shifts to specific areas of improvement, such as reducing visceral fat or increasing skeletal muscle mass. This standardized approach also facilitates better communication between different healthcare providers and systems, as the z-score provides a universal language for risk assessment. As these reference maps are expanded to include more diverse global populations, they will likely become the cornerstone of a new era of precision medicine. The ability to provide an objective, demographic-adjusted assessment of internal health marks a significant departure from the one-size-fits-all approach that has dominated medicine for the last century.
Linking Internal Tissue Distribution to Chronic Conditions
The findings of this large-scale research provided stark evidence regarding the prognostic value of detailed internal tissue mapping. Specifically, the study identified a 2.26-fold increased risk of developing type 2 diabetes for individuals who exhibited high levels of visceral adipose tissue, regardless of their total weight. This reinforces a growing medical consensus that the fat stored around internal organs is metabolically active and highly detrimental to insulin sensitivity. Furthermore, high levels of intramuscular fat were strongly associated with a 1.54-fold increased risk of major adverse cardiovascular events. These insights demonstrate that looking at the internal distribution of fat is far more important than measuring total body mass, as it reveals the specific locations where metabolic dysfunction is most likely to begin. By identifying these high-risk individuals early, healthcare systems can deploy targeted interventions that are far more effective than general weight-loss advice.
On the other end of the spectrum, the research highlighted the critical importance of maintaining skeletal muscle mass for long-term survival and vitality. Individuals who fell into the lowest categories for skeletal muscle scores faced a 1.44-fold higher risk of all-cause mortality, suggesting that muscle preservation is a primary indicator of longevity. This finding shifts the focus of preventive care toward the quality of the tissue rather than just the absence of fat. It suggests that a patient with a high BMI who maintains high muscle mass may actually be at lower risk than a “thin” patient with high internal fat and low muscle density. By quantifying these specific relationships between tissue types and health outcomes, the study proved that AI-driven body mapping can serve as a powerful tool for risk stratification. This approach allows clinicians to move beyond the blunt instrument of the scale and toward a sophisticated understanding of how an individual’s internal composition influences their future health prospects.
Integrating Predictive Imaging into Modern Clinical Workflows
The synthesis of deep learning and large-scale imaging has established a new blueprint for the future of predictive diagnostics and personalized healthcare. While Body Mass Index served as a useful screening tool in the past, the industry recognized that automated AI pipelines provided a far more accurate method for identifying high-risk patients. Organizations began exploring the economic paths required to make MRI-based risk assessment a standard part of routine health screenings, focusing on the long-term cost savings associated with early disease prevention. The validation of these biomarkers in diverse, non-European populations became a priority to ensure that the z-score system remained equitable and effective across all demographics. Technical robustness also improved as software developers worked to ensure that AI models produced consistent results regardless of the MRI scanner brand or software version used, which was essential for widespread clinical adoption.
The transition toward these data-driven insights ultimately encouraged a more proactive approach to patient management and preventive intervention. Medical professionals realized that the distribution and quality of fat and muscle were superior predictors of disease, leading to the development of more specialized treatment plans. Regulatory bodies worked alongside researchers to approve these AI-derived biomarkers for standard medical use, ensuring that the technology met the highest standards of safety and efficacy. Future considerations focused on integrating these automated reports directly into electronic health records, allowing for real-term tracking of a patient’s metabolic trajectory over several years. As the healthcare industry moved away from outdated metrics, the focus remained on using precision tools to save lives through earlier and more accurate diagnoses. This era of medicine prioritized the unique physiological data of each individual, marking a definitive end to the era of generalized weight-based health assessments.