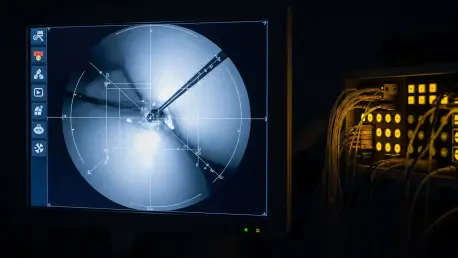
The complexity of modern operating rooms demands a level of vigilance that often pushes the limits of human sensory perception during high-stakes surgical maneuvers. While surgeons possess extraordinary skill, the subtle onset of internal bleeding during robotic or laparoscopic procedures can
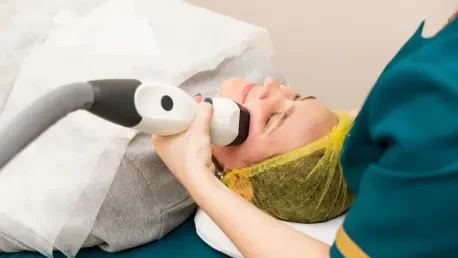
The landscape of modern medical practice is increasingly defined by a relentless tension between clinical excellence and the crushing weight of administrative bureaucracy that threatens to strip physicians of their professional autonomy. As independent dermatology practices navigate the
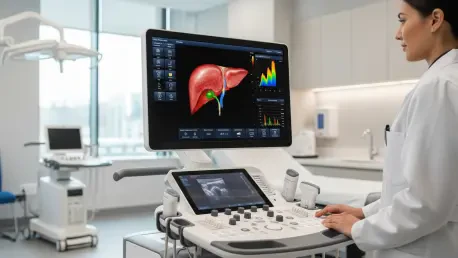
Chronic liver disease currently affects millions of individuals globally, yet the diagnostic journey remains fraught with fragmented procedures that often delay critical interventions and treatment. The introduction of the Mindray Hepatus Series marks a definitive shift toward an integrated

James Maitland brings a unique perspective to the intersection of patient care and cutting-edge technology. As an expert in robotics and IoT applications in medicine, he has watched the healthcare landscape shift from skeptical pilot programs to a reality where artificial intelligence is a daily

The contemporary landscape of American healthcare is currently defined by a relentless struggle to balance high-quality patient care with the suffocating weight of administrative requirements and financial instability. Hospital systems are navigating a period of unprecedented operational pressure,

James Maitland has spent years at the intersection of technology and medicine, witnessing how administrative friction can stall even the most advanced clinical teams. As an expert in streamlining healthcare operations through digital infrastructure and IoT, he understands that the path to patient
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25