The silent progression of osteoporosis often remains undetected until a debilitating fracture occurs, creating a significant burden on global healthcare systems and reducing the quality of life for millions of aging individuals. Standard diagnostic procedures, while effective, are frequently underutilized due to cost or lack of patient awareness, leading to a massive gap between those who have the condition and those who receive a formal diagnosis. However, the vast amount of imaging data already generated during routine medical care presents a unique opportunity to bridge this gap without additional radiation exposure or clinical visits. By leveraging advanced artificial intelligence and machine learning algorithms, medical professionals are now finding ways to extract highly detailed bone health information from standard computed tomography scans originally intended for other purposes. This paradigm shift toward opportunistic screening represents a critical evolution in preventive medicine, turning existing diagnostic workflows into a powerful frontline defense against metabolic bone diseases.
Advanced Computational Analysis: Moving Beyond Traditional Metrics
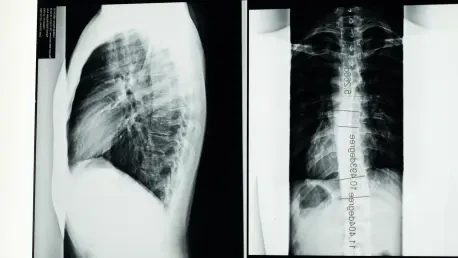
Building on the need for more proactive diagnostic tools, researchers at the Affiliated Jiangning Hospital with Nanjing Medical University recently completed a comprehensive evaluation of machine-learning radiomics models. Their study focused on 166 patients who underwent both dual-energy X-ray absorptiometry and lumbar spine computed tomography, providing a robust dataset for comparing traditional methods against modern computational approaches. By analyzing 656 individual vertebrae from the L1 to L4 levels, the research team utilized three-dimensional segmentation to capture the intricate details of the bone structure. This process allowed for the extraction of 851 distinct radiomic features per region of interest, ranging from basic shape dimensions to complex gray-level co-occurrence matrices. Such a high level of detail far exceeds the capabilities of the human eye, which typically relies on relatively simple visual cues or basic density measurements to assess bone health in a clinical setting.
To ensure the reliability of these findings, the research team implemented a rigorous selection process to narrow the initial feature set down to nine core indicators. These specific indicators were chosen based on their reproducibility and statistical relevance to bone mineral density, focusing on first-order statistics and complex texture-based metrics that reflect the multidimensional microstructure of vertebral bone marrow. Unlike standard Hounsfield unit measurements, which only provide a localized estimate of physical density, these radiomic features offer a window into the underlying biological architecture of the bone. By quantifying the spatial distribution of pixel intensities and the heterogeneity of the tissue, the model identified subtle patterns associated with bone loss that are often missed by traditional imaging interpretation. This transition from qualitative observation to quantitative data analysis marks a significant milestone in the digital transformation of musculoskeletal radiology.
Performance Benchmarks: The Efficacy of XGBoost Models
The central finding of this investigation highlighted the superior performance of the extreme gradient boosting model, commonly referred to as XGBoost, over conventional diagnostic scores. In a direct comparison against vertebral bone quality scores and Hounsfield unit measurements, the XGBoost model achieved an impressive area under the curve of 0.91 within the test cohort. This level of accuracy was supported by a sensitivity of 89 percent and a negative predictive value of 94 percent, suggesting that the model is exceptionally reliable at identifying patients who do not require immediate intervention while flagging those at high risk. Decision curve analysis further validated that this radiomics-based approach provides a higher net clinical benefit than existing methods, offering radiologists a more precise tool for risk stratification. These results indicated that the integration of artificial intelligence into the imaging pipeline could substantially enhance the utility of routine spinal scans.
Despite these superior results, the researchers maintained an objective perspective regarding the current limitations of the technology and its specific role within the broader clinical framework. The proposed model was designed to function as an efficient screening mechanism rather than a total replacement for the current gold standard of dual-energy X-ray absorptiometry. Several challenges remain before widespread adoption can occur, including the retrospective nature of the analysis and the reliance on data from a single institution using a specific type of CT scanner. Furthermore, the current iteration of the model demonstrated limited nuance in stratifying patients specifically diagnosed with osteopenia, a precursor to osteoporosis that requires distinct management strategies. Acknowledging these hurdles is essential for the responsible development of medical AI, ensuring that new tools are both generalizable and reliable across diverse patient populations and hardware configurations.
Clinical Integration: Future Directions for Bone Health Diagnostics
The research team concluded that the path forward required a concentrated effort on multicenter validation to ensure that the radiomics-XGBoost model performed consistently across different healthcare environments. They recommended that future implementations focus on automating the three-dimensional segmentation process, which remained a labor-intensive step in the current workflow. By refining the algorithm to handle varied imaging protocols and noise levels, the technology achieved a state where it could be seamlessly integrated into picture archiving and communication systems. The study emphasized that while the initial findings were groundbreaking, the true value of the technology would be realized only when it provided actionable insights to clinicians in real-time. This involved developing standardized reporting templates that translated complex radiomic signatures into clear, evidence-based recommendations for follow-up care or formal bone density testing.
In addition to technical refinements, the investigators proposed expanding the scope of the model to include a broader range of demographic variables and clinical risk factors. They envisioned a hybrid diagnostic tool that combined radiomic data with patient history and biochemical markers to create a holistic profile of an individual’s skeletal health. This approach aimed to address the current limitations in detecting osteopenia, allowing for earlier intervention and more personalized treatment plans. By shifting the focus from reactive treatment to proactive screening, the healthcare industry positioned itself to reduce the incidence of fragility fractures significantly. The researchers finalized their assessment by noting that the successful deployment of these models depended on interdisciplinary collaboration between radiologists, data scientists, and primary care physicians to ensure that the digital insights translated into tangible patient benefits.