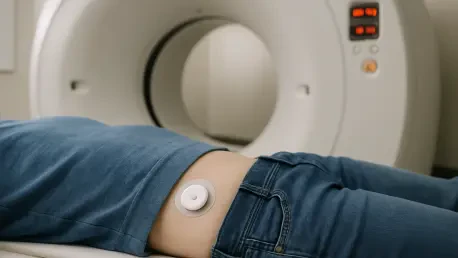
The intersection of diabetes management and diagnostic radiology has long been a source of significant anxiety for patients and healthcare providers alike, often resulting in the unnecessary disposal of expensive medical equipment. As continuous glucose monitors (CGMs) become the standard of care for millions of Americans, the question of whether these skin-attached sensors can survive the rigors of medical imaging has remained largely unanswered by standardized clinical protocols. This ambiguity frequently leads to a “better safe than sorry” approach, where patients are instructed to remove their sensors before any imaging procedure, regardless of the actual risk involved. This practice not only imposes a financial burden on the patient, as a single sensor can cost several hundred dollars, but also interrupts the vital stream of data necessary for managing a chronic and potentially life-threatening condition. Recent research aims to replace this cautious guesswork with a foundation of empirical evidence, offering a more nuanced understanding of how different imaging modalities interact with the electronic and metallic components of modern glucose sensing technology.
Assessing Diagnostic Risks and Equipment Integrity
Differentiating Between Ionizing Radiation and Magnetic Fields
The primary challenge in establishing safety guidelines for CGMs lies in the fundamental physical differences between various medical imaging technologies and how they interact with sensor hardware. Research led by Brittany Gundersen, a specialist in radiation sciences at Virginia Commonwealth University, has meticulously categorized these interactions to dispel common misconceptions. Standard X-rays and Computed Tomography (CT) scans utilize ionizing radiation, which passes through the body to create images of internal structures. The study confirmed that while these high-energy waves are powerful, they do not possess the specific physical properties required to damage the internal circuitry of a CGM or cause it to malfunction during a standard diagnostic session. Consequently, the prevailing fear that radiation exposure will “fry” the delicate electronics of a glucose monitor is largely unfounded, provided the exposure remains within standard clinical parameters used for routine diagnostic purposes.
Building on this discovery, the research highlights that the integrity of the device remains intact after exposure to X-ray or CT radiation, meaning the sensors can continue to provide accurate readings once the scan is complete. This finding is particularly significant because it challenges the widespread institutional policy of mandatory sensor removal for all radiological procedures. By distinguishing between the harmless nature of ionizing radiation and the more volatile nature of magnetic resonance, clinicians can now provide patients with more accurate advice. This shift in perspective ensures that the continuity of glucose monitoring is only interrupted when a genuine physical threat to the hardware or the patient exists. This evidence-based distinction serves as the cornerstone for new hospital protocols that aim to balance diagnostic necessity with the practical realities of daily chronic disease management.
Evaluating the Impact of Image Artifacting on Diagnostics
While the physical safety of the device during an X-ray or CT scan is now better understood, the research also addresses the secondary concern of image quality, specifically the phenomenon known as artifacting. Because CGMs contain metallic components and high-density plastic, they can cause visual distortions on the resulting medical images, appearing as bright spots or streaks that might obscure the underlying anatomy. The study found that while these artifacts are present, they are generally localized to the area immediately surrounding the sensor site. In many cases, if the area being imaged is distal to the sensor—for example, a chest X-ray while the sensor is on the upper arm—the artifact poses no threat to the diagnostic utility of the scan. Radiologists can often work around these visual “noise” elements without requiring the patient to strip away their monitoring equipment.
However, the research notes that the decision to leave a sensor in place during an X-ray or CT scan should still involve a brief consultation between the technologist and the interpreting physician. If the sensor is positioned directly over the specific area of clinical interest, such as a suspected bone fracture or a suspicious mass, the artifacting might indeed interfere with a precise diagnosis. In these specific instances, removal becomes a matter of diagnostic clarity rather than hardware safety. By understanding that the risk is “visual” rather than “functional,” healthcare teams can make more informed, case-by-case decisions. This approach prevents the reflexive disposal of sensors when the imaging site is far removed from the device, thereby saving patients money and reducing the psychological stress associated with frequent sensor changes and the subsequent recalibration periods.
Protocol Development for Magnetic Resonance Environments
Understanding the Hazards of High-Strength Magnetic Resonance
In contrast to the relatively benign nature of X-rays, Magnetic Resonance Imaging (MRI) presents a vastly different set of risks due to the powerful static magnetic fields and radiofrequency pulses used to generate images. The research confirms that CGMs, which contain ferromagnetic materials and conductive wires, are strictly contraindicated for use inside the MRI bore. The magnetic field can exert significant physical force on the metallic components, potentially dislodging the sensor from the skin or causing internal mechanical failure. Furthermore, the radiofrequency energy can induce electrical currents in the sensor’s circuitry, leading to rapid heating of the needle or filament embedded under the patient’s skin. This poses a direct risk of thermal burns, making it an issue of immediate physical safety rather than just equipment longevity or image quality.
The electronic failure resulting from MRI exposure is often catastrophic and irreversible, frequently leading to total device death or, more dangerously, the reporting of highly inaccurate glucose values. The study emphasizes that the sophisticated algorithms used by CGMs to translate chemical signals into glucose data can be easily corrupted by the intense electromagnetic environment of an MRI suite. Because the stakes are so high—ranging from patient injury to incorrect medical dosing based on false readings—the research reinforces the necessity of removing all CGM components before entering the magnetic field. This clarity is essential for radiology departments to maintain a safe environment, ensuring that both the patient and the expensive imaging equipment are protected from the unintended consequences of ferromagnetic interference during the scanning process.
Implementing Evidence-Based Patient Education and Preparation
The translation of these research findings into clinical practice requires a proactive approach to patient communication and scheduling. Gundersen’s work suggests that the most effective way to manage the intersection of CGMs and medical imaging is through advanced notification and education. Patients should be screened for the presence of a CGM at the time of scheduling, rather than at the time of the appointment, to allow for proper planning. For an MRI, patients must be advised to bring a replacement sensor and the necessary supplies to re-apply the device after the procedure is finished. This foresight prevents the frustration of a cancelled appointment or the sudden loss of glucose monitoring capability without a backup plan, which can be particularly distressing for those with Type 1 diabetes who rely heavily on real-time data.
Moreover, the research highlights the importance of empowering radiographers and nurses with specific technical knowledge so they can reassure patients undergoing standard X-rays. By explaining the difference between the “safe” radiation of an X-ray and the “hazardous” magnetism of an MRI, providers can build trust and reduce patient anxiety. This educational shift also involves coordinating with insurance providers and device manufacturers to ensure that sensors removed for medical necessity are more easily replaced. The ultimate goal of this research is to create a clinical environment where technology and diagnostics coexist harmoniously. By applying these specific findings to daily operations, medical facilities can transition away from outdated, blanket policies toward a sophisticated model of care that respects the patient’s technological needs while ensuring the highest standards of diagnostic safety and accuracy.