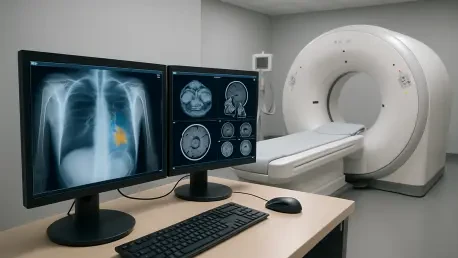
The evolution of medical imaging has reached a critical juncture where traditional anatomical snapshots are no longer sufficient to meet the demands of early-stage disease detection and personalized therapy. While standard magnetic resonance imaging has provided decades of invaluable structural data, its inherent sensitivity limitations often leave metabolic changes—the earliest indicators of pathology—shrouded in digital noise. By harnessing the principles of quantum physics, specifically through the manipulation of nuclear spins, researchers have unlocked a way to illuminate these hidden biological processes. This paradigm shift is being led by innovations that bridge the gap between abstract quantum computing theories and the tangible needs of clinical diagnostics. The ability to observe cellular metabolism in real-time offers a profound window into living tissue, potentially identifying aggressive cancers or neurological shifts long before physical tumors or lesions become visible to the naked eye through conventional scans.
Harnessing Hyperpolarization for Enhanced Diagnostics
The core of this technological leap lies in the application of hyperpolarized hydrogen to transform common metabolic intermediates into highly sensitive super-markers for clinical use. Using the proprietary POLARIS platform, the process amplifies the signal generated by MRI scanners by more than 10,000-fold, effectively turning a whisper into a shout within the diagnostic environment. This methodology allows for the tracking of molecules like pyruvate as they move through the body and are processed by cells, providing a vivid map of metabolic activity. Unlike traditional methods that rely on radioactive tracers, this quantum-based approach uses stable isotopes, making it inherently safer for repeated patient monitoring. The level of detail achieved through this signal enhancement enables physicians to observe the exact moment a cell begins to malfunction, which is crucial for oncology and cardiovascular research where timing determines the success of an intervention.
Integrating such sophisticated quantum technology into existing hospital infrastructure represents a significant operational advantage because it does not require a complete overhaul of current MRI hardware. Building on this foundation, the technology acts as a plug-and-play enhancement that utilizes the massive installed base of scanners already present in medical facilities worldwide. This scalability ensures that the benefits of quantum-enhanced imaging are not confined to a few elite research institutions but can eventually become a standard of care in regional hospitals. By focusing on the metabolic biomarkers that drive disease progression, this approach naturally leads to a more nuanced understanding of patient-specific responses to treatment. The efficiency of this system allows for faster data acquisition, which reduces the time patients spend inside the scanner while simultaneously increasing the volume of actionable information provided to the oncology or neurology team.
Strategic Infrastructure and Market Integration
The transition from theoretical research to practical application has been fueled by a strategic expansion of research hubs and collaborative networks across the globe. A significant milestone was achieved through the establishment of a dedicated research center at the University of Cambridge, which now serves as a primary European axis for quantum-enhanced metabolic imaging. This facility operates in tandem with ongoing projects at prestigious locations like the Memorial Sloan Kettering Cancer Center, creating a robust pipeline for sharing data and refining diagnostic protocols. These partnerships are essential for validating the technology across diverse patient populations and ensuring that the metabolic insights gained are consistent and reproducible. The presence within the Cambridge Innovation Cluster allows for a unique synergy between radiology experts and quantum physicists, fostering an environment where technical breakthroughs are immediately tested against clinical needs.
Financial and academic support has played a pivotal role in maintaining the momentum of these developments, with a noticeable shift in the profile of modern technology investors. Interestingly, much of the capital sustaining these advancements has come from venture firms specializing in quantum computing and deep tech rather than traditional pharmaceutical or medical device backgrounds. This influx of funding, including a substantial international round of thirty million dollars, has enabled the workforce to double and the operations to move into specialized facilities in major science parks. Under leadership that prioritizes the intersection of physical sciences and healthcare, the focus has expanded from preclinical small-animal studies to the rollout of products designed specifically for human applications. This growth trajectory highlights a broader industry trend where quantum technology is no longer viewed as a futuristic concept but as a current, viable solution for complex medical challenges.
Clinical Trajectory and Future Patient Outcomes
The movement toward human-scale applications marked a definitive shift in how clinicians approached the management of chronic and life-threatening conditions. In the period from 2026 to 2028, the focus transitioned toward integrating these quantum-enhanced tools into routine diagnostic workflows for patients suspected of having early-stage neurodegenerative diseases. By utilizing the super-marker capabilities of the POLARIS platform, medical teams successfully bypassed the limitations of traditional protein-based markers, which often appeared too late for effective intervention. This proactive stance allowed for the implementation of neuroprotective therapies at a stage when cellular damage was still reversible. Furthermore, the ability to monitor real-time metabolic response to chemotherapy provided a mechanism for adjusting dosages instantly, ensuring that patients received the most effective treatment with the least amount of systemic toxicity, thereby improving overall quality of life.
Practical implementation of these quantum diagnostics required a multifaceted approach involving specialized training for radiologists and the establishment of new regulatory frameworks. Stakeholders worked to standardize the production of hyperpolarized agents, ensuring that every facility had access to the necessary materials to perform high-sensitivity scans. Moving forward, the industry should prioritize the democratization of this technology by simplifying the hyperpolarization process further to reduce operational costs. Organizations must also look toward expanding the library of available metabolic markers to include those specific to metabolic disorders like diabetes or fatty liver disease. The successful deployment of these tools demonstrated that the synergy between quantum physics and medicine was not merely a research curiosity but a transformative force that redefined the boundaries of what was possible in non-invasive patient diagnostics.